

Greek patients with Cystic Fibrosis (CF) are registered in:
- the European Cystic Fibrosis Society Patient Registry (ECFSPR) of the European Cystic Fibrosis Society (ECFS)
- in the National Registry of Cystic Fibrosis Patients of our country of the Ministry of Health
Patients’ Registries collect demographic and clinical data, which is pseudonymised and anonymised.
Patients’ registries are useful and valuable tools that contribute to better patient care with the ultimate goal of improving patients’ health .
Moreover, Patients’ Registries give the chance to patients to take part in clinical trials of new medicines.


European Cystic Fibrosis Society Patient Registry- ECFSPR
Το Ευρωπαϊκό Μητρώο Κυστικής Ίνωσης ECFSPR- European Cystic Fibrosis Society Patient Registry δημιουργήθηκε το 2008 από την Ευρωπαϊκή Επιστημονική Κοινότητα Κυστικής Ίνωσης- ECFS.
The ECFSPR is linked to the National Cystic Fibrosis Registry of Greece and collects pseudonymised demographic and clinical data from more than 54,000 Cystic Fibrosis patients from 40 participating European countries. The ECFSPR reflects the reality of the disease across Europe.
In 2010, the Cystic Fibrosis Department of the General Hospital “Hippocrates” was the first Greek C.I. Centre to join the European Registry. After many efforts in continuous cooperation between the Association and the Scientific Committee of our country, we managed for the first time in 2015 to include all Greek Cystic Fibrosis Centres in Thessaloniki and Athens in the European Registry.
The inclusion of our country in the European Register of C.I. has increased the prestige, credibility and dynamics of our country. Also in this way Greece contributed to the joint efforts of the European Cystic Fibrosis community to register patients.
The data collected by each CF Center in our country is pseudonymised and sent once a year to the European Cystic Fibrosis Patient Registry (ECFSPR). The data is collected with the consent of the patients or their parents in the case of minor patients.
The information collected by the ECFS is used to measure, research and compare aspects of Cystic Fibrosis and its treatment in participating countries, to deepen understanding of the disease, to improve standards of care, to provide data for epidemiological research and to facilitate public health planning.
Benefits of Greece’s participation in the European Cystic Fibrosis Registry (ECFSPR)
- Evaluating the effectiveness of various treatments of each CF Centre and comparing them with those of other CF Centres in other countries in Europe and around the world.
- Assessing which treatments are most useful and whether they improve patient care.
- Assessment of the epidemiology of the disease, e.g. complications, new infectious strains.
- Designing future innovative medicines for CF patients.
- Identifying groups of patients eligible for new treatments, ensuring access to innovative therapies.
- Apart from the scientific community, the data of the ECFSPR is also used by patient representatives for the planning and documentation of the Association’s positions and demands in order to improve the health and quality of life of patients.
Useful links
- Detailed information on the ECFSPR: https://www.ecfs.eu/ecfspr
- Detailed annual reports of ECFSPR: https://pr.ecfs.eu/annual-reports/
- Updated Information and Consent Form: here
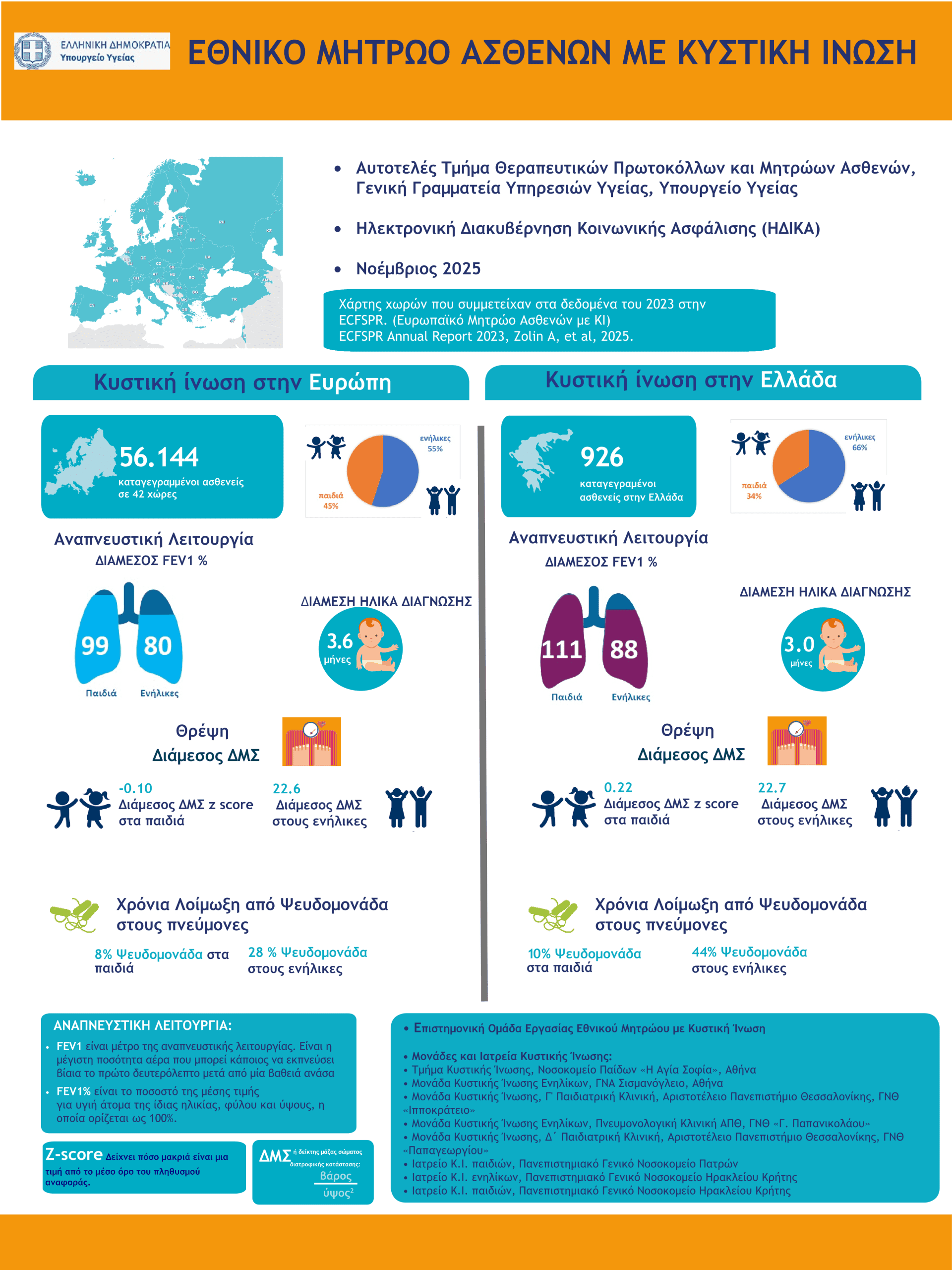
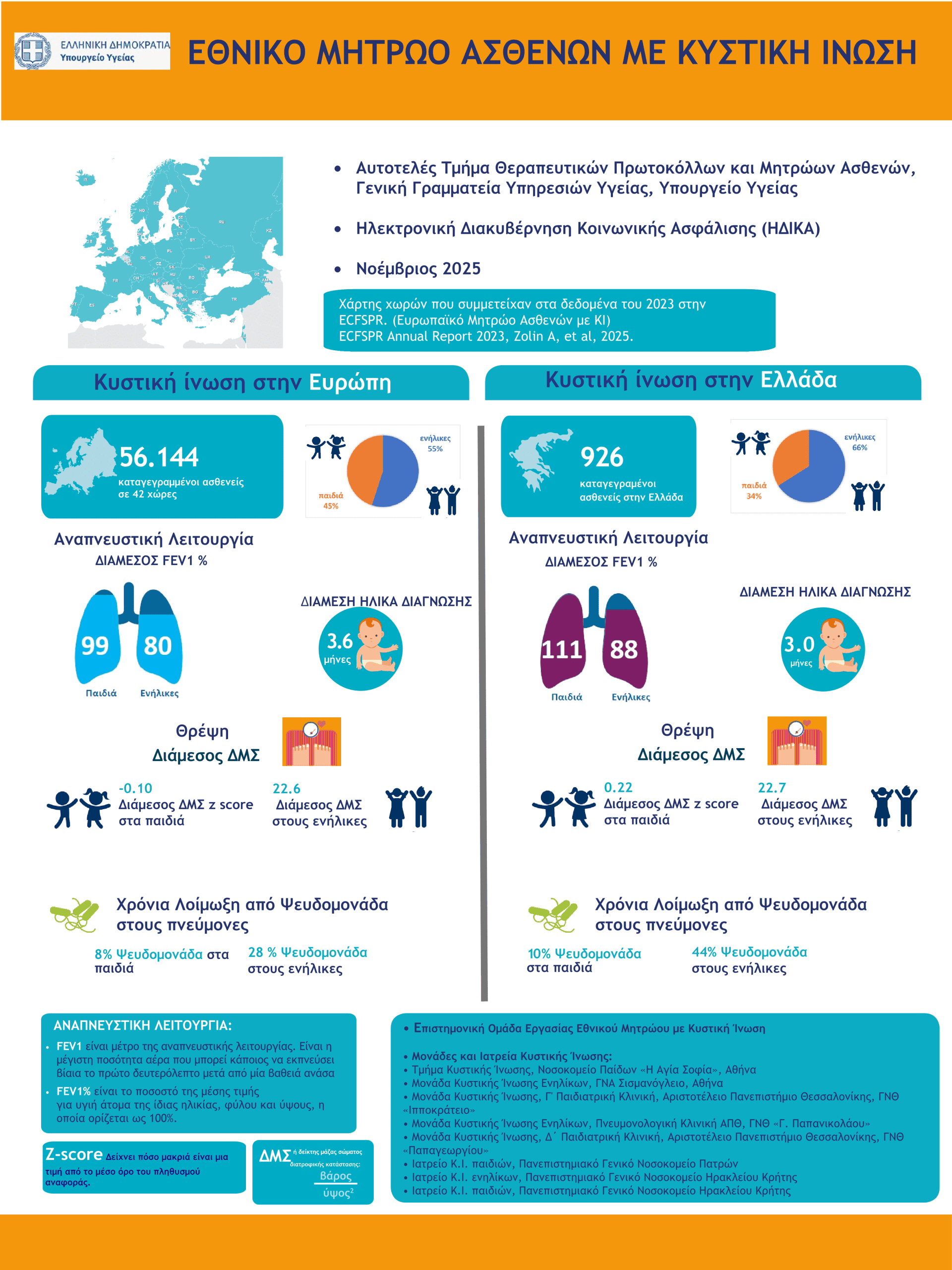
National Cystic Fibrosis Patients' Registry
The National Cystic Fibrosis Registry of Greece was established as a priority in 2020 by the Ministry of Health (here the Government GazetteIt is one of the first patient registries in Greece and is linked to the European Cystic Fibrosis Registry ECFSPR .
It is A key tool for the provision of Health Services to Greek patients with C.I. The The establishment of the National Register was necessary prerequisite for the completion of the reimbursement negotiation procedures for CFTR Modulators revolutionary treatments in Greece. The registration of Greek Cystic Fibrosis patients in the National Registry of CF is mandatory based on a decision of the Ministry of Health (here).
Responsible for carrying out the processing and storage of the data of patients in the National Register, on behalf of Ministry of Health, is EDIKA SA. The management and supervision of the National Register is carried out by the Independent Department of Treatment Protocols and Patient Registers of the Ministry of Health.
The Data Protection Officer (DPO) of the Ministry of Health, as the controller, monitors the compliance of the establishment and operation of the Registry for the protection of patients’ personal data in cooperation with the DPO of IDIKA SA. The monitoring, processing and periodic updating of the National Register is carried out by the Working Group, which consists of members of the Scientific Committee and a patient representative of the Association.
Infogpraphics of National CF Registry of Greece
The first infographics of Greek patients with Cystic Fibrosis in Greece based on the data of the National CF Registry was published in 2023 by the Greek Ministry of Health.
- Greek Ministry of Health infographics for 2023: here
- Greek Ministry of Health infographics for 2024: here
- Greek Ministry of Health infographics for 2025: here
Working Group Members of the National CF Patients’ Registry
- DIAMANTEA FILIA, Pulmonologist, Director of the Cystic Fibrosis Unit for Adults, “SISMANOGLEIO” General Hospital
- DOUROS KONSTANTINOS, Professor of Pediatrics – Pediatric Pulmonology, 3rd Pediatric Clinic of the University of Athens, P.G.H. “ATTIKON”
- THIRAIOS ELEFTHERIOS, General Practitioner/Family Physician, Director of H.S.Y., Head of the General Directorate of O.D.I.P.Y. S.A., Secretary General of the Athens Medical Society
- KARETSI ELENI, Pulmonologist, Director of E.S.Y., University General Hospital of Larissa
- LOUKOU IOANNA, Pediatrician, Director of the Cystic Fibrosis Department, Athens Children’s Hospital “Aghia Sophia”
- MANIKA AIKATERINI, Associate Professor of Pulmonology – Tuberculosis, University of Thessaloniki, Adult Cystic Fibrosis Centre, Department of Pulmonology, University of Thessaloniki, General Hospital “G. PAPANIKOLAOU”
- PANAGIOTA MITROU, Specialist Pathologist – Diabetologist, Doctor of the University of Athens, Head of the Independent Department of Treatment Protocols and Patient Registers of the Ministry of Health
- BOLIS KONSTANTINOS, Pediatrician, Coordinating Director of the Pediatric Department, University Pediatric Clinic, Pediatric Pulmonology Department, University General Hospital of Patras “PANAGIA I VOTHIEIA”
- PARASKAKIS EMMANOUL, Associate Professor of Pediatrics, Head of the Pediatric Pulmonology Unit, Pediatric Clinic of PAGNI, President of the Hellenic Pediatric Pulmonology Society
- PITSIDIANAKIS GEORGIOS, Pulmonologist – Tuberculologist, Director of the Cystic Fibrosis Clinic for Adults and Bronchiectasis, Pulmonology Clinic, Heraklion University General Hospital (PAGNI)
- SPINOU ANNA, Patient Advocate – EUPATI fellow, President of the Panhellenic Cystic Fibrosis Association, Member of the Board of EURORDIS- Rare Diseases Europe, ePAG- ERN LUNG
- TSANAKAS IOANNIS, Professor Emeritus of Pediatric Pulmonology, Aristotle University of Thessaloniki, Aristotle University of Thessaloniki, AUTH
- FOTOULAKI MARIA, Professor of Pediatrics – Pediatric Gastroenterology, Director of the 4th University Pediatric Clinic of the Aristotle University of Thessaloniki, “PAPAGEORGIOU” Hospital, Head of the Cystic Fibrosis Unit of the Intersectoral Cystic Fibrosis Center of the Aristotle University of Thessaloniki.
- HATZIAGOROU ELPIDA, Professor of Pediatrics – Pediatric Pulmonology, Aristotle University of Thessaloniki, Head of the Cystic Fibrosis Unit, 3rd Pediatric Clinic, GP “IPOKRATEIO”
The coordinator of the Working Group is TSANAKAS IOANNIS.
